 |
Audio Asylum Thread Printer Get a view of an entire thread on one page |
For Sale Ads |
 |
Audio Asylum Thread Printer Get a view of an entire thread on one page |
For Sale Ads |
108.67.99.125
In Reply to: RE: Toyota poised to unleash another revolution, just like the Prius was. posted by tinear on November 19, 2014 at 07:05:31
Why natural gas, of course. The frackers are going to love this but will environmentalists, presumably the intended market, feel the same? And filling station infrastructure? Since the Prius made use of an existing energy infrastructure, at least it didn't have an inconvenience or range anxiety hurdle to overcome so comparison of it to a fuel cell vehicle is not really valid in terms of potential market acceptance.
Overall this whole thing strikes me as typical LA auto show press release hyperbole...here today, gone tomorrow. We shall see.
Follow Ups:
Need a pretty prodigious energy source to drive THAT, though.
Maybe "we" can use some magic combination of solar, geothermal, tidal, and wind energy to power water electrolysis.
I'm still pullin' for nuclear fusion ("harnessing the power of the stars") myself.
... and I am STILL waitin' for my jetpack!
:-)
all the best,
mrh
Is xylose.
It can be 'digested' using enzymes at low temperature (50C) with nearly 100% efficiency.
Alternatively one could use photocatalytic water splitting.
This is a form of artificial photosynthesis which splits water into its constituent parts using solar panels.
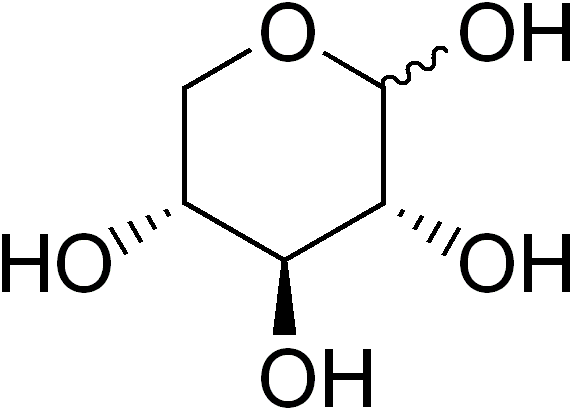
and there's the problem -- the structure of xylose contains C-H bonds (which oxidize with the release of some respectable amount of energy, akin to our much-beloved hydrocarbons) -- but there're too many hydroxyl groups in a carbohydrate to get a lot of heat energy out per molecule by combustion -- compared to petroleum products, are mostly hydrocarbon: CH3(CH2)nCH3 e.g., (long-chain aliphatic hydrocarbon).
The good thing, of course, is that xylose is readily renewable (so-called "wood sugar", and, yes, it does grow on trees, so to speak) but pound per pound, not so great really.
In the interest of full and complete disclosure - xylose is an aldopentose; C5H10O5, but it does have that meaningless but compelling "hydrate of carbon" empirical formula that we think of when we think of classical carbohydrates.
all the best,
mrh
I wasn't thinking of using xylose to burn but as a source of H2.
Xylose can be used to produce hydrogen very efficiently at low temperatures and normal atmospheric pressure by using enzymes to 'digest' it.
a thermophile of some sort?
I could always actually look it up... but I am with you now.
If the bioreactor could be rigged to deliver adequate hydrogen in real time, that'd be very sweet (no pun intended)!
One of the byproducts would, presumably be CO2, though, I assume.
all the best,
mrh
From wiki:Xylose
In 2014 a low-temperature 50 �C (122 �F), atmospheric-pressure enzyme-driven process to convert xylose into hydrogen with nearly 100% of the theoretical yield was announced. The process employs 13 enzymes, including a novel polyphosphate xylulokinase (XK).[84][85]It is a bio-technological process which mimics digestion in an actual living thing.
Works with any kind of plant material. Seems to be quite simple and elegant really.
There is more in the link to Virginia Tech who came up with it all.Then again it could all be a belated April's Fool thing...
Edits: 11/22/14

It's clever and efficient -- but, like normal biological oxidation of carbohydrate, it produces water and CO2.
... oh, and, when I was in school, the single most abundant carbohydrate in Nature was reputed to be glucose (the monomeric building block of starch and cellulose, as well as an extremely important energy source for all cellular metabolism... and half of that tasty disaccharide sucrose!).
I am not a biofuels guy... but I am a carbohydrate biochemist.
all the best,
mrh
the difference to fossil fuels is that those release CO2 which has been stored and taken out of circulation for millions of years while this process cannot release more CO2 than has been fixed in the plant material over the last year or so depending on the crop used.
And the amount released will be fixed again when growing crops to replace the Hydrogen fuel used.
So logically over time the net CO2 released must be close to zero unless you chop down forests and replace them with deserts which would clearly be insane.
-- and compare it to an alkyl (straight-chain) hydrocarbon.
While I see your point, I am not sure on a molar basis that it's gonna be such a great deal. An improvement no doubt, but still, perhaps, a crutch akin to using natural gas (which is seen by many as a crutch) as an energy source.
Interesting topic, for sure!
all the best,
mrh
...the fusion would be used to generate electricity which in turn would be used to generate mass quantities of H2. H2 would become an energy transmission medium using relatively low tech but reliable and terrorism resistant pipeline technology. Fuel cells in the home for electricity. Burn it for heat. Use it for transportation with fuel cells or advanced IC engines. Use it as a chemical and fertilizer feedstock. We would then transition away from a hydrocarbon economy and into a hydrogen economy...for maybe a hundred years or so...fusion isn't as clean as we're sometimes led to believe. But I'm not holding my breath. Practical, industrial grade fusion technology has been just 20 yrs away for the last 60 or so years. IOW, you'll have your jetpack before I have my fusion based hydrogen.
It should be clean, though -- a wee bit o' tritium (radioactive hydrogen isotope) is needed to get the ball rolling, as I understand it... and/or there is some tritum produced by fusion of hydrogen to helium, but tritium is a low-energy beta emitter and reasonably short-lived. Tritiated water isn't too harmful, either... basically you'd whizz it out if ingested. Tritium incorporated covalently into, say, DNA precursor nucleotides might be a bit more of a real-world health risk... but, hey? What's life with out a little risk?!
The only real danger with fusion is to use the Edward Teller method to start the ball rollin'... our thermonuclear bombs use a controlled fissin reaction to collapse a (plutonium!) sphere containing deuterium ('heavy hydrogen'; a stable, nonradioactive hydrogen isotope with both a proton and a neutron in its nucleus) and/or hydrogen -- thus getting the temperature high enough to create a plasma dense enough to begin nuclear fusion.
In fact, as I understand it, the plutonium was used, at least in part, to make sure that the H-bombs were in fact dirty enough to do more than just real-time explosive damage.
Fusion really is pretty clean (too clean for Doc Teller, I think); the power comes from the tiny (on a per-molecule basis) atomic mass deficit between two hydrogen nuclei and one helium nucleus. That itty-bitty bit of mass becomes energy according to Uncle Albert's much-beloved E = m*c^2, where "c" is the speed of light (a pretty big number: 3 x 10^8 m/sec).
all the best,
mrh
...due to an absence of transmission facilities and/or excessive distance from load centers. Some of those locations could be excellent, however, for large-scale production of hydrogen via electrolysis.
+1
Cheers,
Al
Post a Followup:
| FAQ |
Post a Message! |
Forgot Password? |
|
||||||||||||||
|
||||||||||||||
This post is made possible by the generous support of people like you and our sponsors: